Introduction by Pida Ripley, Founder, AGEofthePHAGE.com and CombatAMR.org

I developed an enduring interest in the development and use of phage therapy over two decades ago when I was implementing humanitarian and development programmes in the collapsed states of the former USSR. Working in the Caucasus, I became aware of the bacteriophage therapy work undertaken by the Eliava Institute of Bacteriophages, Microbiology and Virology in Georgia, as many of my Tbilisi staff members used phage therapy rather than antibiotics and one staffer’s husband was a microbiologist at the Institute. I was fascinated to learn that Felix d’Herelle had played an important role in supporting George Eliava and the early development of the Eliava Institute!
In 2006, when back in the UK and about to undergo a total knee replacement and having a long history of multiple serious infections, I made sure I had the full details of the Institute - just in case I ended up with a drug resistant infection in my knee. Thankfully my notes were unnecessary!
However, as I learned more about the bacteriophage, I developed a deep interest in the development of phage therapy. Having survived four potential killer infections during my lifetime, the increasing threat posed by antimicrobial resistance became a major concern. As so few members of the public appeared to be aware of the global AMR threat, I created the CombatAMR.org website - a free global resource to help raise awareness. AGEofthePHAGE.com has been created to support and promote the revival of phage development.
As so few members of the public appeared to be aware of the very real and rapidly advancing drug-resistance infections global threat to our lives, I created and sponsor the CombatAMR.org website - a free global resource to help raise awareness of the relentless increase of antimicrobial resistance (AMR) already stealing too many lives. AGEofthePHAGE.com has been created to support and promote the revival of phage therapies and development of phage applications that will combat AMR and save lives. I am delighted to be helping increase global awareness of the phage with the launch of #WorldPhageDay, 22nd October and #WorldPhageWeek, 22nd-28th October. This time the phage will not retreat into the shadows again.
THE PHAGE - A DEVOURER OF BACTERIA
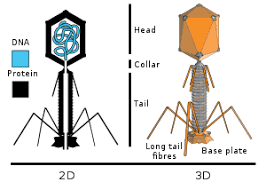
A bacteriophage is a virus that infects and replicates within a specific bacterium. The term bacteriophage is derived from "bacteria" and the Greek: (phagein), "to devour". A specific class of viruses able to infect and destroy bacteria, they are among the most common and diverse entities in the biosphere. Fortunately phages offer no threat to humans. Bacteriophages are composed of proteins that encapsulate a DNA or RNA genome, and may have relatively simple or elaborate structures. Their genomes may encode as few as four genes, and as many as hundreds of genes. Phages replicate within the bacterium following the injection of their genome into the cytoplasm of the bacteria.
They are intrinsically narrow spectrum agents and are restricted to a single bacterial species. Widely distributed in locations populated by bacterial hosts, such as soil or the intestines of animals, one of the most dense natural sources for phages and other viruses is sea water; up to 70% of marine bacteria may be infected by phages.
Despite the desperate clinical battle against infections in the pre-antibiotic era, phage therapy was largely undeveloped by the time penicillin was made widely available in the 1940s. The easy to use and highly effective broad spectrum penicillin antibiotic became known as the 'miracle drug' - saving millions of lives and Western interest in the emerging phage treatment waned. However, phage therapy continued being developed and has been used for over 100 years as an alternative to antibiotics and became a treatment of choice for many in the former Soviet Union and Central Europe, as well as in France.
A problem of this underdeveloped therapy is that very few controlled, well-designed clinical trials have tested its efficacy and safety. The anecdotal only evidence of its success has severely limited wider global implementation, despite successful outcomes for 'last resort' patients and continuing support from populations able to access phage therapy. Validation of phage treatment for human infectious diseases must be obtained by conducting robust clinical trials.
As antimicrobial resistance increases, with no new antibiotics in the pipeline, more governments are beginning to allocate funding for further research on phage therapy. The dramatic advances in technology are enabling the pioneering research required to fully discover the potential of phage therapies and application.
A REVIVAL OF INTEREST IN PHAGE THERAPY
Due to profligate overuse and misuse of antibiotics, bacterial pathogens are developing multi-drug resistance to all our known antibiotic treatments. Antimicrobial resistance (AMR) is increasing at a frightening rate and is being described as a 'ticking time bomb' that poses an "apocalyptic" threat to public health. It is a global problem that must be tackled on a global basis as there is no safe harbour. Rich or poor, young or old - everyone is at risk. If we fail to combat AMR it is predicted that by 2050, drug-resistant infections could be killing more than 10 million people worldwide every year.
During the past three decades the World Health Organisation (WHO) issued multiple warnings about increasing drug resistance, but very little action was taken by governments or by pharmaceutical companies. Many pharmaceutical companies have closed their antibiotic research and development programmes due to irrecoverable high costs involved in seeking new antibiotics. As our 'last line' antibiotics are beginning to be ineffective against multi-drug resistant bacteria, belatedly there is realisation that there are no new antibiotics in the pipeline.
A Global Discovery Gap
We are standing on the threshold of a return to the deadly pre-antibiotic era and urgent global collaborative action is of increasingly critical importance to global human, animal and environmental health. A race is on to search and develop both new antibiotics and alternative therapies. Even if new forms of antibiotics are discovered, there remains a ‘discovery gap’ as the required safety trials take 10-15 years before a new drug is made available.
One of the promising options is the long-overlooked phage therapy. The global community need to be informed about the potential offered by life-saving phage therapies and public dialogue on the potential benefit of investing in phage research and development encouraged. As antibiotic resistance rises across the world, people are individually turning to phage therapy to effectively treat chronic health conditions and life-threatening antibiotic-resistant infections.
Phage therapy cannot provide the total answer to antimicrobial resistance
but it certainly is an essential complementary therapy.


